[DSE化學2019]同學準備好DSE CHEM未?
化學艾維斯
1001 回覆
153 Like
27 Dislike
amide高d 因爲計返多d lone pair / H available for Hbond
compare ionic lattice唔考啦 都無學過
2007 ce q10
點解ethyl ethanoate 唔可以用polystyrene cup 裝著?
點解ethyl ethanoate 唔可以用polystyrene cup 裝著?

想問下ce 1985 39
點解個titration冇ppt form都可以用electrical conductivity黎determine end-point?


點解個titration冇ppt form都可以用electrical conductivity黎determine end-point?



ethyl ethanoate係organic solvent
polystyrene應該會溶到係solution入面?
咁隻杯咪會爛
polystyrene應該會溶到係solution入面?
咁隻杯咪會爛

ester relatively non polar
PS also non polar
會溶左你個膠杯
PS also non polar
會溶左你個膠杯
都係睇total concentration of mobile ion
題目係acid-base titration 冇比實數 淨係話small portions of acid successively added
係咪當form出黎既h2o唔導電?
係咪當form出黎既h2o唔導電?
G持
當你一路add acid to alkaline直到剛好equivalence point (all H+ OH- reacted to give H2O) 相比你過左equivalence point繼續add excess acid 個total concentration of mobile ion會上升 咁你就會見到個明顯既轉角位
另一個原因係H+ / OH-外既ion 對conductivity既contribution係會低d
所以for左H2O後 雖然有其餘ion喺mixture 但都會睇到conductivity跌 只係無BaSO4(s)果d case咁明顯

另一個原因係H+ / OH-外既ion 對conductivity既contribution係會低d
所以for左H2O後 雖然有其餘ion喺mixture 但都會睇到conductivity跌 只係無BaSO4(s)果d case咁明顯


 thx
thx


呀 sir 唔好意思我又想再問下
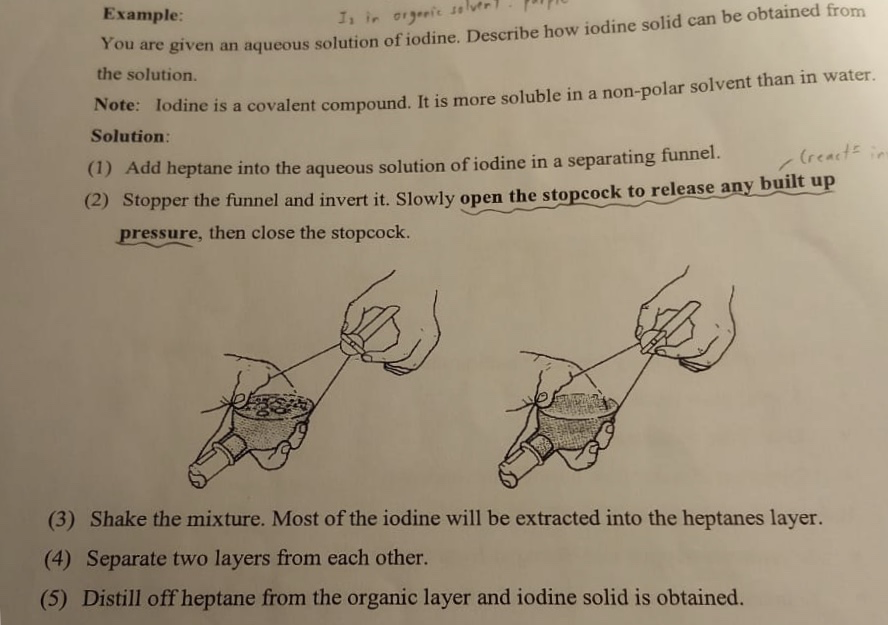
佢就話如果想obtain iodine solid, 就先用liquid liquid extraction , 再distill off organic solvent
咁點解我唔一開始就直接煲走d水?

佢就話如果想obtain iodine solid, 就先用liquid liquid extraction , 再distill off organic solvent
咁點解我唔一開始就直接煲走d水?
iodine volatile
要煲走水iodine都會走曬
要煲走水iodine都會走曬
巴打今年仲有冇執試題分佈個張list?



Assume 2A(g) + B(s) <-> C(g)
如果加B會唔會影響個eqm position ?
如果加B會唔會影響個eqm position ?
有 應該下個禮拜會拍片
唔會 solid concentration係constant (assume change of surface area can be neglected)
oh thx

咁extract 左organic solvent 就唔volatile ?



咁即係如果一個全部都aq嘅equilibrium ,係咁加水落去都唔會郁到eqm position ,因為水個濃度都係constant?
organic solvent boiling point可以好低 例如ether講緊35 oC 個yield loss咪細d
水自己濃度constant
但多左水會dilute左其他species 咁d數會唔同左
要準確就要要plug d新數入去試
但多左水會dilute左其他species 咁d數會唔同左
要準確就要要plug d新數入去試
1001
